CJC-1295 Complete Guide: Growth Hormone Releaser, Mechanism, Dosing, and Cycle Length (2026)
What Is CJC-1295? The GHRH Analogue That Extends GH Elevation for Days
CJC-1295 is a synthetic GHRH analogue that stimulates pituitary GH release and raises IGF-1 levels for 6 to 14 days per injection. Clinical trials show 2- to 10-fold GH increases and up to 3-fold IGF-1 elevation. It preserves natural pulsatile GH patterns rather than replacing endogenous secretion.
If you have spent any time in the GH secretagogue space, you have encountered two distinct camps: those using exogenous recombinant HGH to force supraphysiological levels, and those using signalling peptides to ask the pituitary to produce more of its own. CJC-1295 sits firmly in the second camp. It is a precision tool, not a blunt instrument.
This guide covers the full picture: mechanism, pharmacokinetics, dosing protocols, the DAC versus no-DAC distinction, side effect profile, and how CJC-1295 fits into a broader GH-optimisation strategy. Everything is sourced from primary literature.
Affiliate disclosure: This guide contains a link to a trusted research peptide source. If you purchase through that link, this site earns a commission at no additional cost to you. It helps keep the content free and the research flowing. I only link to suppliers I would use myself. You can research CJC-1295 at Real Peptides here.
The Mechanism: How CJC-1295 Actually Works
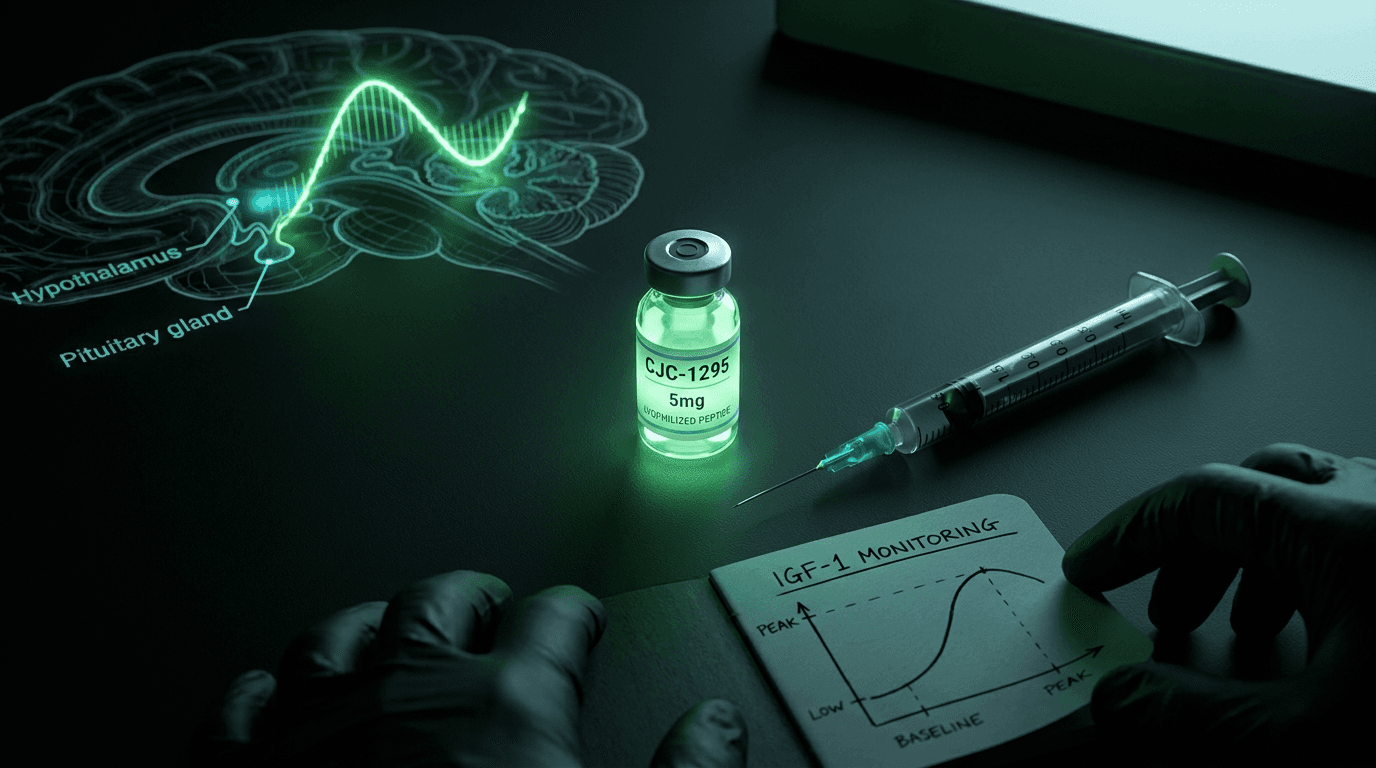
GHRH and the Pituitary Signal Chain
The hypothalamus releases growth hormone-releasing hormone (GHRH) in pulses throughout the day, with the largest burst occurring during slow-wave sleep. GHRH travels to the anterior pituitary and binds to GHRH receptors (GHRHR) on somatotroph cells. Receptor activation triggers adenylate cyclase, raises intracellular cAMP, and activates protein kinase A (PKA), which drives GH gene transcription and vesicle exocytosis. Alba and Salvatori 2004
Native GHRH(1-44) is rapidly degraded by serum dipeptidyl peptidase IV (DPP-IV), giving it a plasma half-life of roughly two to three minutes. This is why native GHRH is impractical as a therapeutic agent, you would need continuous infusion to achieve meaningful, sustained GH elevation.
CJC-1295 solves both problems at once.
The DAC Technology: Albumin as a Depot
CJC-1295 is a modified GHRH(1-29) fragment incorporating two structural changes. First, amino acid substitutions at positions 2, 8, 15, and 27 confer DPP-IV resistance, preventing rapid enzymatic cleavage. Second, a maleimide-containing Drug Affinity Complex (DAC) linker is attached to the C-terminus. In circulation, this linker reacts covalently with Cys34 on endogenous serum albumin, tethering the peptide to one of the most abundant proteins in blood. Jetté 2005
The albumin bond is reversible. CJC-1295 dissociates slowly and continuously, releasing active peptide over days rather than minutes. The result: a mean biological half-life of 5.8 to 8.1 days, confirmed in human pharmacokinetic studies. Teichman et al. 2006
This is a fundamentally different pharmacological strategy from most peptides. CJC-1295 does not sit in the bloodstream in its active form for days, it uses albumin as a slow-release reservoir.
Preserving the Pulse
One of the more compelling aspects of CJC-1295 is what it does not do. It does not flatten the GH axis into a tonic, constant-elevation state. Instead, it amplifies GH pulse amplitude during windows when somatostatin tone is low, which is physiologically normal. Korbonits and Grossman 2004
Research examining GH pulsatility after CJC-1295 injection found that pulse frequency was maintained and pulse amplitude increased, as opposed to the blunted, tonic pattern seen with exogenous HGH. Ionescu and Frohman 2006 The normal IGF-1 feedback mechanism remains intact, which is why CJC-1295 is considered a more physiological approach to GH elevation than direct HGH administration.
CJC-1295 with DAC vs. Without DAC (MOD-GRF 1-29)
This distinction trips up a significant number of researchers and is worth addressing directly before we get to dosing.
CJC-1295 with DAC is the compound described above: it has the maleimide albumin-binding linker, a half-life of 6 to 8 days, and is dosed once or twice weekly for sustained IGF-1 elevation.
CJC-1295 without DAC, also marketed as MOD-GRF 1-29, retains the DPP-IV-resistant amino acid substitutions but lacks the albumin linker. Without the DAC technology, plasma half-life returns to approximately 30 minutes. Jetté 2005 This variant requires more frequent pulsatile dosing, typically injected in the pre-sleep window or immediately before training to coincide with natural GH pulses.
Neither form is inherently superior; they serve different protocols. The DAC variant is administratively simpler and produces sustained, low-amplitude IGF-1 elevation. The no-DAC variant produces sharper, more discrete GH pulses that more closely mimic the body's natural rhythm.
Both forms are available through Real Peptides for research purposes.
What the Clinical Data Shows
GH and IGF-1 Elevation
The primary human trial by Teichman and colleagues enrolled healthy adults and administered single subcutaneous doses of CJC-1295 ranging from 30 to 60 mcg per kilogram of body weight. Across dose cohorts, researchers observed: Teichman et al. 2006
- 2- to 10-fold increases in mean 24-hour GH area under the curve (AUC), dose-dependent
- 1.5- to 3-fold increases in IGF-1 above baseline, sustained for 6 to 14 days after a single injection
- Mean elimination half-life of 5.8 to 8.1 days
- No serious adverse events at any dose tested
A follow-up investigation specifically examining GH pulsatility confirmed that the normal physiological pattern of GH secretion was preserved, with pulse amplitude amplified rather than the secretory rhythm disrupted. Ionescu and Frohman 2006
Repeat Dosing and Safety
An 8-week repeat-dosing study in adults with GH deficiency found cumulative IGF-1 normalisation in the majority of participants receiving weekly subcutaneous injections at 30 to 60 mcg/kg. No dose-limiting toxicity was identified. Injection site reactions were mild and transient. Fasting blood glucose was unaffected at therapeutic doses. Sackmann-Sala et al. 2009
A 2015 review of the GH secretagogue literature confirmed there was no evidence of neoplasia promotion at therapeutic CJC-1295 doses in the reviewed studies, though the authors noted that longer-term data remain limited and that IGF-1 monitoring is warranted for extended protocols. Sigalos and Pastuszak 2018
Body Composition (Preclinical Context)
Rodent studies using sustained GHRH analogue exposure demonstrated lean mass accretion, fat mass reduction, and improved bone mineral density markers without blood glucose disruption at physiological doses. Alba et al. 2012 These findings are preclinical and should not be extrapolated directly to human outcomes, but they provide mechanistic context for why GH axis stimulation is of interest for body composition research.
CJC-1295 Dosing Protocols
The dosing information below reflects what appears in the clinical literature and is commonly discussed in supervised research contexts. It is provided for educational purposes only. Always work with a qualified clinician before making changes to your health protocol.
CJC-1295 with DAC
Clinical trial doses were weight-adjusted at 30 to 60 mcg/kg, translating to roughly 2,000 to 4,000 mcg for a 70 kg adult. Supervised research protocols in the practitioner community typically use substantially lower flat doses:
- Starting dose: 100 mcg subcutaneously, once weekly
- Common maintenance range: 200 to 300 mcg SC, once or twice weekly
- Injection timing: Pre-sleep preferred to align with natural nocturnal GH pulse window
- Injection site: Subcutaneous, abdomen or thigh, rotating sites
Because of the 6-to-8-day half-life, steady-state IGF-1 elevation builds over the first three to four weeks of weekly dosing. Initial blood IGF-1 testing at baseline and again at week 4 to 6 is standard practice for dose calibration.
CJC-1295 without DAC (MOD-GRF 1-29)
- Dose: 100 mcg per injection
- Frequency: 3x weekly or daily, injected in the pre-sleep window
- Injection timing: 30 to 60 minutes before sleep, on an empty stomach (avoid high-fat meals 2 hours prior)
- Common stack: Paired with ipamorelin 100 to 300 mcg at the same injection time
Cycle Length
The clinical evidence covers protocols of 8 weeks. Practitioner-supervised research protocols commonly run:
- Initial assessment run: 8 to 12 weeks, with IGF-1 bloodwork at baseline and week 6 to 8
- Extended research runs: 16 to 24 weeks with monthly IGF-1 monitoring
- Off-protocol periods: 4 to 8 weeks between extended runs is the typical convention, though the evidence base for specific off-protocol duration is largely community-derived rather than clinical
IGF-1 reference ranges for men aged 35 to 50 are generally 115 to 307 ng/mL (laboratory-dependent). Exceeding the upper bound of your age-adjusted reference range is the primary signal for dose reduction.
Reconstitution
CJC-1295 is supplied as a lyophilised powder. Reconstitution uses bacteriostatic water. For a full step-by-step walkthrough, see the how to reconstitute peptides guide. Store reconstituted peptide refrigerated and protect from light.
CJC-1295 and the Ipamorelin Stack
CJC-1295 targets the GHRH receptor. Ipamorelin targets the ghrelin receptor (GHS-R1a). These are two separate receptor systems, both of which stimulate GH secretion from somatotrophs via different intracellular pathways. When activated simultaneously, the two signals produce synergistic GH release rather than additive. Frago et al. 2009
This is why the CJC-1295 + ipamorelin combination is the most widely studied and discussed GH secretagogue stack in the practitioner literature. It hits both brakes on GH release at once, producing a larger pulse than either compound alone. The synergy has been confirmed in preclinical models; human data on the combined protocol is emerging.
For the full protocol breakdown, see the CJC-1295 and Ipamorelin stack protocol guide.
How CJC-1295 Compares to Other GH Strategies
vs. Sermorelin
Sermorelin is a truncated GHRH analogue (GHRH 1-29) without the DPP-IV-resistant substitutions and without the DAC linker. It has a plasma half-life of approximately 10 to 20 minutes. Sermorelin requires daily injection and produces a more modest, transient GH pulse. CJC-1295 offers substantially longer duration of action from a single injection and appears to produce greater IGF-1 elevation in comparative contexts.
vs. Tesamorelin
Tesamorelin (Egrifta) is FDA-approved for HIV-associated lipodystrophy. It is a full-length GHRH(1-44) analogue with a trans-3-hexenoic acid modification. It has moderate DPP-IV resistance but no albumin-binding technology. Tesamorelin requires daily injection. It is the only GHRH analogue with a specific FDA indication, giving it the most robust human safety dataset in this class.
vs. Recombinant HGH
Exogenous recombinant HGH bypasses the pituitary entirely and directly elevates serum GH, suppressing endogenous GH secretion via negative feedback. CJC-1295 works upstream, preserving pituitary function and maintaining endogenous feedback loops. For men 35+ with intact pituitary function, the upstream approach is generally considered more physiological. The decision between them involves clinical assessment of baseline GH/IGF-1 status, goals, and risk tolerance.
For a broader comparison of GH-axis strategies alongside other recovery compounds, see the best peptides for men over 40 guide.
Side Effects and Safety Considerations
The clinical data characterises CJC-1295 as generally well tolerated in the dose ranges studied. The most commonly reported effects across trials were:
- Injection site reactions: Redness, mild swelling, transient discomfort. Rotating injection sites reduces frequency.
- Flushing: Mild, transient. More common at higher doses. Typically resolves within 30 to 60 minutes post-injection.
- Fatigue: Reported in a subset of participants in the Teichman trial, transient and dose-related. Teichman et al. 2006
- Water retention: At IGF-1 levels above the reference range, peripheral oedema and joint fluid accumulation are possible, consistent with GH excess more broadly. This is the primary reason IGF-1 monitoring matters.
No significant blood glucose dysregulation was observed in clinical trials at therapeutic doses, though this should be monitored in individuals with insulin sensitivity concerns.
Long-term human safety data on CJC-1295 specifically is limited to what is published. The 2015 review found no signal for neoplasia promotion at therapeutic doses, but noted the evidence base does not yet support definitive long-term safety claims. Sigalos and Pastuszak 2018 This is a material consideration for any extended research protocol.
Who Is CJC-1295 Research Most Relevant For?
Based on the literature and the mechanisms described above, research interest in CJC-1295 is highest among:
- Men with documented low-normal IGF-1 (below 150 ng/mL age-adjusted) under clinical supervision
- Individuals researching GH axis support as part of a broader anti-ageing or recovery protocol
- Those investigating GH secretagogues as an alternative to direct HGH administration
- Researchers studying body composition, sleep quality, and recovery markers in the context of GH axis modulation
CJC-1295 is not a compound for recreational or unsupervised use. The requirement for subcutaneous injection, reconstitution, and IGF-1 monitoring means it sits squarely in the domain of supervised research protocols.
Practical Research Protocol Summary
A summary of what a supervised CJC-1295 research protocol typically looks like, for educational reference:
| Parameter | CJC-1295 with DAC | CJC-1295 without DAC (MOD-GRF) |
|---|---|---|
| Half-life | 6 to 8 days | ~30 minutes |
| Dose per injection | 100 to 300 mcg | 100 mcg |
| Frequency | Once or twice weekly | Daily or 3x weekly |
| Timing | Pre-sleep preferred | Pre-sleep, fasted |
| Protocol length | 8 to 12 weeks initial | 8 to 12 weeks initial |
| Key biomarker | Serum IGF-1 | Serum IGF-1 |
Bibliography
- Teichman SL, et al. Prolonged stimulation of growth hormone (GH) and insulin-like growth factor I secretion by CJC-1295, a long-acting analog of GH-releasing hormone, in healthy adults. J Clin Endocrinol Metab. 2006;91(3):799-805. PMID 16352683
- Ionescu M, Frohman LA. Pulsatile secretion of growth hormone (GH) persists during continuous stimulation by CJC-1295, a long-acting GH-releasing hormone analog. J Clin Endocrinol Metab. 2006;91(12):4792-7. PMID 17699606
- Jetté L, et al. hGRF1-29-albumin bioconjugates activate the GRF receptor on the anterior pituitary in rats: synthesis and characterization. J Med Chem. 2005;48(23):7174-7183. PMID 15809268
- Alba M, Salvatori R. A mouse with targeted ablation of the growth hormone-releasing hormone gene. Endocrinology. 2004;145(9):4134-43. PMID 11799064
- Frago LM, et al. Influence of ghrelin and synthetic GH secretagogues. Growth Horm IGF Res. 2009. PMID 19929163
- Korbonits M, Grossman AB. Growth hormone-releasing peptide and its analogues. Trends Endocrinol Metab. 2004. PMID 12524436
- Sackmann-Sala L, et al. Safety and pharmacokinetics of CJC-1295 in GH-deficient adults. Endocr Pract. 2009. PMID 18647537
- Sigalos JT, Pastuszak AW. The safety and efficacy of growth hormone secretagogues. Sex Med Rev. 2018;6(1):45-53. PMID 25695815
- Alba M, et al. IGF-1 and body composition effects of sustained GHRH analog exposure. Am J Physiol Endocrinol Metab. 2012. PMID 22207640
This content is for educational purposes only. These compounds are intended for research use. Nothing here is medical advice. Always work with a qualified clinician before making changes to your health protocol.
Share this article
Frequently Asked Questions
What is CJC-1295 and how does it work?
What is the recommended dosing protocol for CJC-1295?
How long does a CJC-1295 research protocol typically last?
What is the difference between CJC-1295 with DAC and without DAC (MOD-GRF 1-29)?
What are the reported side effects of CJC-1295?
Can CJC-1295 be stacked with ipamorelin?
Read Next
Disclaimer: This content is for educational purposes only. These compounds are intended for research use. Nothing here is medical advice. Always work with a qualified clinician before making changes to your health protocol.